ABSTRACT
Although there is evidence that the ratio of 2nd to 4th digit length (2D:4D) correlates with prenatal testosterone level, psychological and health traits only a single study has assessed the relationship with brain morphological features. Here we investigated the association between the 2D:4D ratio and several brain subvolumes. Seventy-five subjects between the ages of 18 and 30 were included in the study. The length of the 2nd and 4th digits were measured with an electronic vernier caliper while MRI measurements were performed on a Siemens Magnetom Trio Tim (3 T) system. Freesurfer software suite was used for volumetric segmentation. Finger ratio significantly correlated with total cerebral cortex, total cerebellar white matter and total cerebellar cortex in males but not in females. Our results indicate that prenatal testosterone, as estimated by the 2D:4D ratio has an effect on adult brain morphology in males.
Gergely Darnai1*, Enikő Plózer1, Gábor Perlaki1,2,3, Gergely Orsi1,2,3, Szilvia Anett Nagy2, Réka Horváth1, Attila Schwarcz3,4, Norbert Kovács1, Anna Altbäcker1,5 József Janszky1,3 and Zsófia Clemens1,6
1Department of Neurology, University of Pécs, Pécs, Hungary
2Pécs Diagnostic Centre, Pécs, Hungary
3MTA-PTE Clinical Neuroscience MR Research Group, Pécs, Hungary
4Department of Neurosurgery, Pécs, Hungary
5Institute of Cognitive Neuroscience and Psychology, Research Centre for Natural Sciences, Hungarian Academy of Sciences, Budapest, Hungary
6National Institute of Clinical Neuroscience, Budapest, Hungary
INTRODUCTION
Testosterone is known to shape neuronal connectivity in the developing brain [1]. There is evidence [2] that prenatal testosterone level correlates with the ratio of 2nd to 4th digit length (2D:4D). Given that the 2D:4D fixes in utero and the ratio remains stable during development [3] it is regarded as a prenatal marker of testosterone exposure. The 2D:4D has consistently been shown to be sexually dimorphic with males showing lower finger ratios than females [3,4]. The 2D:4D ratio has been related to a set of psychological, cognitive, behavioral, morphological and health traits. Low digit ratio (that is more androgenized hands) is associated with aggression [5] and less empathy [6] in males and assertiveness in females [7]. Those with more androgenized hands (that is with low 2D:4D) have higher spatial and sporting ability. Despite the abundance of cognitive studies so far only a single few studies have assessed the relation between finger ratio and a brain morphological feature, the asymmetry of hippocampal subregions [8] and the volume of the dorsal anterior cingulate cortex [9]. Here we assessed the relationship between the 2D:4D ratio and brain subvolumes including intracranial volume, total cerebral white matter, total cerebral cortex, total ventricular volume, total cerebellar white matter and total cerebellar cortex in healthy university students.
METHODS
Subjects
Seventy-five subjects (32 males and 43 females) between the ages of 18 and 30 were included in the study. Mean age (± SD) was 23.4 (±3.3) in males and 22.9 (±2.7) in females. All subjects were Caucasian university students without a history of brain disorders, drug or alcohol abuse as also assessed by a structured questionnaire. The study was approved by the local ethical committee of the University of Pécs and all subjects gave written informed consent.
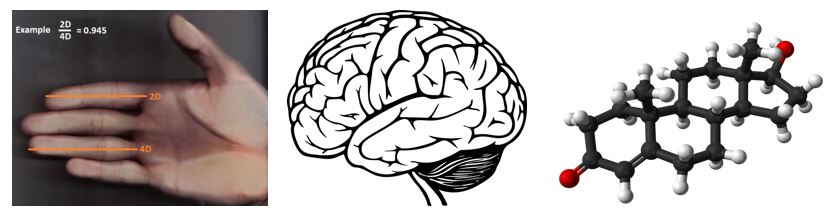
Measurement of 2D:4D finger ratio
The length of the 2nd and 4th digits was measured directly using an electronic vernier caliper (precision: 0.01 mm) because this method minimizes the possibility of distortion [10]. Fingers were measured on both hands twice from the basal crease of each finger to the tip of the finger. The digit ratio calculated for the two measurements were averaged. Then an average was calculated for the right and left hands.
Magnetic resonance imaging
Magnetic resonance imaging (MRI) measurements were performed on a Siemens Magnetom Trio Tim (3 T) system (Siemens AG, Erlangen, Germany), with a 12-channel head coil. For the volumetric analysis a T1 weighted sagittal magnetization-prepared rapid acquisition with gradient echo (MPRAGE) images were used: field of view = 256 × 2562 mm, repetition time= 2530 ms, echo time = 3.37 ms, TI = 1100 ms, slice thickness = 1 mm, slice number = 176, flip angle=7°, bandwidth = 200 Hz/pixel, 256 × 256 matrix.
MRI data post processing
Freesurfer 4.5.0 software suite was used for cortical reconstruction and volumetric segmentation of the images (http://surfer.nmr.mgh.harvard.edu/). Freesurfer software can be used for reliable automated brain segmentation for structures and allows us to assess the volume of the pre-defined brain structures. Freesurfer’s semi-automatic anatomical processing steps were executed on all subjects’ data including removal of the non-brain tissue, the automated Talairach transformation, volumetric segmentation, intensity normalization, tessellation of borders between the grey matter and white matter surfaces and using intensity gradients to localize the grey matter, white matter, and fluid boundaries. Accuracy of the Talairach transformation, skull strip, white matter and pial surfaces and subcortical segmentation were visually checked in case of every subject after each script. Error correction was applied where it was necessary. The final segmentation is based on a probabilistic atlas and the subject-specific measured values. The following structures were used for statistical analysis: total intracranial volume (ICV), total cerebral white matter (TCWM), the left hemisphere cortical grey matter and right hemisphere cortical grey matter summarized as total cerebral cortex (TCC), total cerebellar white matter (TCeWM) and total cerebellar cortex (TCeC), total ventricular volume (TVV) which was the sum of the left and right lateral ventricle, left and right inferior lateral ventricle, third and fourth ventricles.
Statistical calculations
Spearman Rank-Order Correlation between finger ratio and the six subvolumes were calculated for males and females separately. In a next step multiple linear regression models were constructed to control for age in those bivariate correlations that were found significant.
RESULTS
There was no significant gender difference (p=.183) between males [mean (SD) = .998 (.021)] and females [mean (SD) = 1.008 (.039)] in the 2D:4D ratio. The intra class correlation coefficients between the first and second measurements indicated a good level of reliability both in males (.90) and females (.98).
In males finger ratio significantly correlated with TCC (rs=.45; p=.01), TCeC (rs=.423; p=.016) and TCeWM (rs=.364; p=.041). A statistical control for age did not essentially change the correlation coefficients (see Table 1). A positive correlation trended toward significance between finger ratio and ICV in males (rs=.333; p=.063).
No significant correlation was found in females however there was a tendency toward a negative correlation between finger ratio and ICV (rs=-.281; p=.068).
Table 1. Univariate and multivariate linear regression models estimating the association of 2D:4D finger ratio and brain subvolumes in A) males and B) females. One asterisk denotes p < 0.05, two asterisks denote p < 0.01. Abbreviations: ICV – intracranial volume, TCC – total cerebral cortex, TCWM – total cerebral white matter, TVV – total ventricular volume, TCeC – total cerebellar cortex, TCeWM – total cerebellar white matter.
DISCUSSION
Our results indicate a positive association between finger ratio reflecting low prenatal testosterone concentration and volume of the cerebral cortex, total cerebellar cortex and total cerebellar white matter in males. Prenatal testosterone is known to shape neuronal connectivity and to affect postna-tal psychological and behavioral traits [11]. Effects of testosterone on the nervous system are, how-ever, manifold and differ according to the species studied, region of the brain, neuronal cell type and timing of exposure with regard to critical periods of neurodevelopment [12].
The positive correlation between finger ratio and increased volume of the cerebral cortex is in line with results from human neuronal cell cultures where elevated testosterone level was shown to induce cell loss by inducing apoptosis [13]. In a longitudinal study both positive and negative correlations with fetal testosterone levels were found for specific regions of cortical gray matter [1]. The reported positive correlations include bilateral somatosensory, motor and premotor cortices [1].
In our sample no gender difference in digit ratio was found. From a previous study it is known that digit ratio is associated with faculty membership of academics [14]. Therefore it seems plausible that the absence of gender difference is due to the predominance of both male and female subjects from the same faculty, namely the medical faculty. Average digit ratio of ~ 1.00 found in our sample for both genders corresponds with the female norm of ~ 1.00 also found for science faculty students irrespective of gender in the study by Brosnan [14].
From animal studies it is known that prenatal testosterone may have differing effect on neurodevelopment in males and females [15]. Yet, given the sparse data on humans, we cannot explain why prenatal testosterone index in our study is associated with cerebral cortex volume in males only.
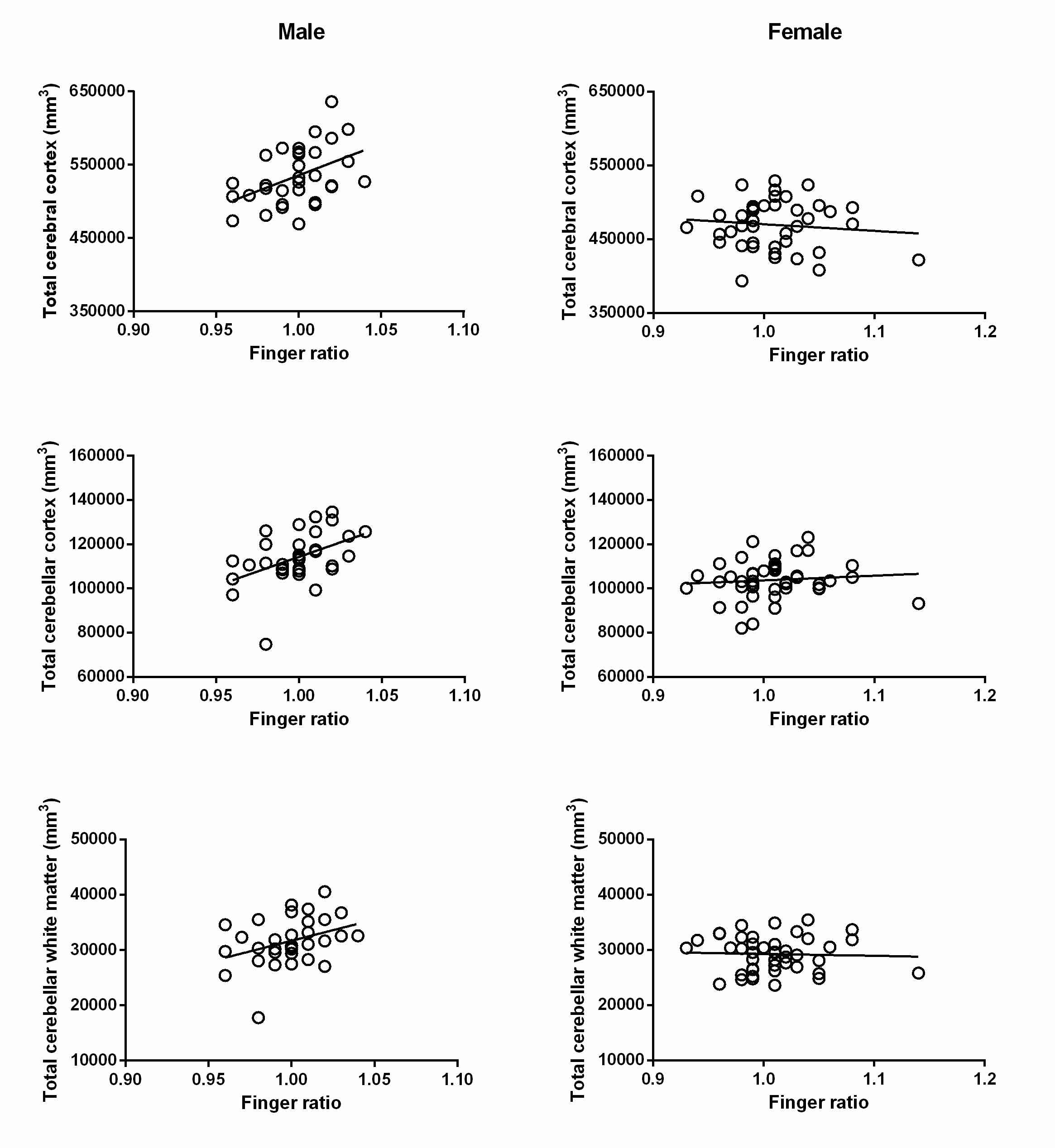
Figure 1. Scatterplots representing the relationships between finger ratio and specific brain volumes (total cerebral cortex, total cerebellar cortex and total cerebellar white matter) in males and females separately. The correlations in males remained significant after correction for age.
CONCLUSION
To our knowledge this is the second study assessing the relation between 2D:4D and brain MRI features and the first assessing the association between 2D:4D and gross anatomical measures of the human brain. Given that 2D:4D is considered as a reliable estimate of prenatal testosterone, future studies assessing additional brain structures may reveal a more detailed picture about regional morphological effects of prenatal testosterone.
REFERENCES
[1] M. V Lombardo, E. Ashwin, B. Auyeung, B. Chakrabarti, K. Taylor, G. Hackett, et al., Fetal testosterone influences sexually dimorphic gray matter in the human brain., The Journal of neuroscience. 32 (2012) 674–680. doi:10.1523/jneurosci.4389-11.2012.
[2] S. Lutchmaya, S. Baron-Cohen, P. Raggatt, R. Knickmeyer, J.T. Manning, 2nd to 4th digit ratios, fetal testosterone and estradiol, Early Hum. Dev. 77 (2004) 23–28. doi:10.1016/j.earlhumdev.2003.12.002.
[3] J.T. Manning, D. Scutt, J. Wilson, D.I. Lewis-Jones, The ratio of 2nd to 4th digit length: a predictor of sperm numbers and concentrations of testosterone, luteinizing hormone and oestrogen., Hum. Reprod. 13 (1998) 3000–3004. doi:10.1093/humrep/13.11.3000.
[4] Z. Zheng, M.J. Cohn, Developmental basis of sexually dimorphic digit ratios., Proc. Natl. Acad. Sci. U. S. A. 108 (2011) 16289–94. doi:10.1073/pnas.1108312108.
[5] A.A. Bailey, P.L. Hurd, Finger length ratio (2D?: 4D) correlates with physical aggression in men but not in women, Biol. Psychol. 68 (2005) 215–222. doi:10.1016/j.biopsycho.2004.05.001.
[6] A. Von Horn, L. BÄckman, T. Davidsson, S. Hansen, Empathizing, systemizing and finger length ratio in a Swedish sample, Scand. J. Psychol. 51 (2010) 31–37. doi:10.1111/j.1467-9450.2009.00725.x.
[7] G.D. Wilson, Finger-length as an index of assertiveness in women, Pers. Individ. Dif. 4 (1983) 111–112. doi:10.1016/0191-8869(83)90061-2.
[8] J. Kallai, Á. Csathó, F. Kövér, T. Makány, J. Nemes, K. Horváth, et al., MRI-assessed volume of left and right hippocampi in females correlates with the relative length of the second and fourth fingers (the 2D:4D ratio), Psychiatry Res. - Neuroimaging. 140 (2005) 199–210. doi:10.1016/j.pscychresns.2004.05.010.
[9] A.X. Gorka, R.E. Norman, S.R. Radtke, J.M. Carré, A.R. Hariri, Anterior cingulate cortex gray matter volume mediates an association between 2D:4D ratio and trait aggression in women but not men., Psychoneuroendocrinology. 56 (2015) 148–56. doi:10.1016/j.psyneuen.2015.03.004.
[10] J.T. Manning, B. Fink, N. Neave, N. Caswell, Photocopies yield lower digit ratios (2D:4D) than direct finger measurements., Arch. Sex. Behav. 34 (2005) 329–33. doi:10.1007/s10508-005-3121-y.
[11] B. Auyeung, M. V Lombardo, S. Baron-Cohen, Prenatal and postnatal hormone effects on the human brain and cognition., Pflugers Arch. 465 (2013) 557–71. doi:10.1007/s00424-013-1268-2.
[12] A.P. Arnold, R.A. Gorski, Gonadal steroid induction of structural sex differences in the central nervous system., Annu. Rev. Neurosci. 7 (1984) 413–42. doi:10.1146/annurev.ne.07.030184.002213.
[13] M. Estrada, A. Varshney, B.E. Ehrlich, Elevated testosterone induces apoptosis in neuronal cells, J. Biol. Chem. 281 (2006) 25492–25501. doi:10.1074/jbc.M603193200.
[14] M.J. Brosnan, Digit ratio and faculty membership: implications for the relationship between prenatal testosterone and academia., Br. J. Psychol. 97 (2006) 455–466. doi:10.1348/000712605X85808.
[15] J.A. Morris, C.L. Jordan, S.M. Breedlove, Sexual differentiation of the vertebrate nervous system., Nat. Neurosci. 7 (2004) 1034–1039. doi:10.1038/nn1325.
 Rehabilitáció csak online elérhető
Rehabilitáció csak online elérhető
 E-mail: paleomedicina@gmail.com
E-mail: paleomedicina@gmail.com